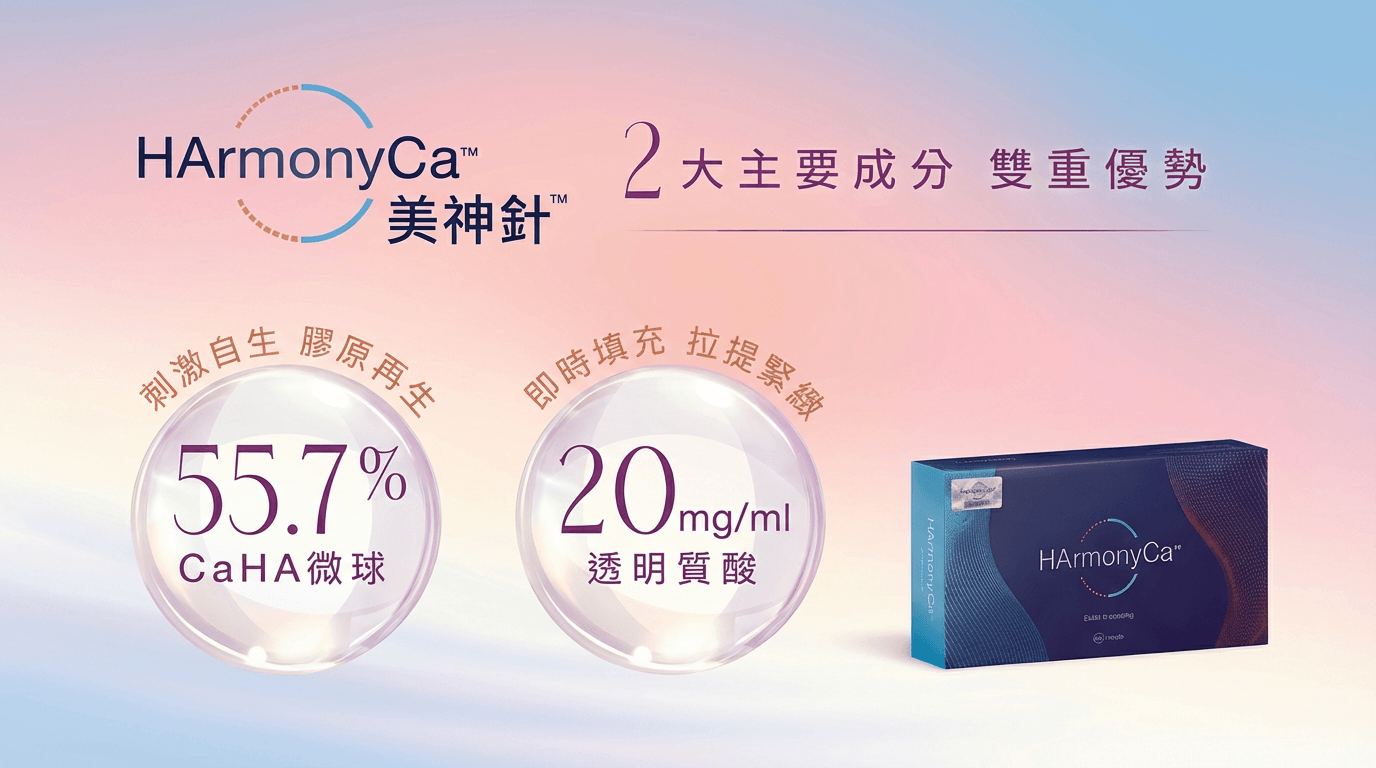

HArmonyCa™ 美神針™ 全新雙效療程

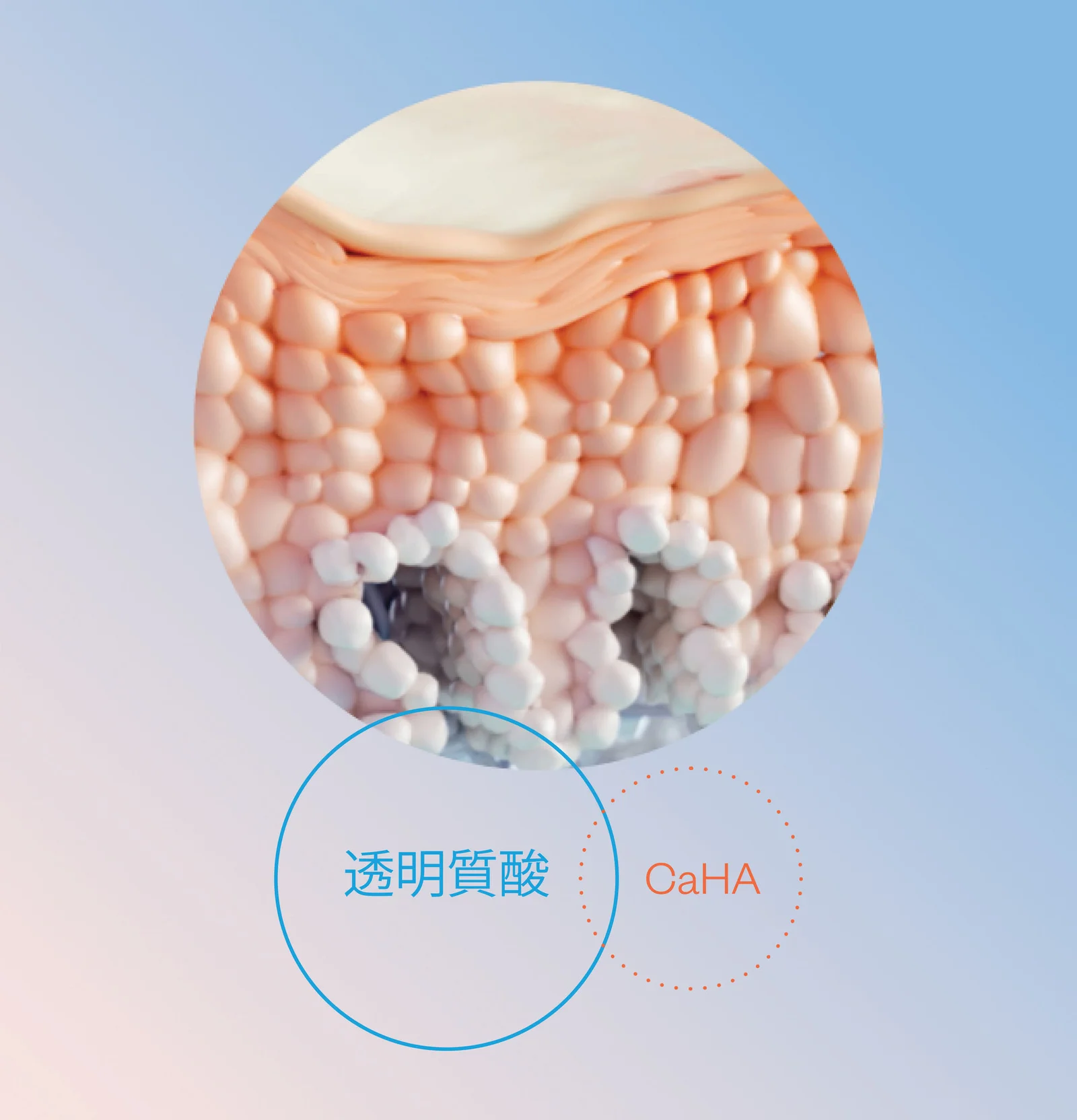
膠原蛋白讓您的肌膚重現豐盈感

療程效果展示

臨床研究證實效果
臨床研究顯示,91.6%受試者
整體外觀獲得改善‡‡
180天後確認治療區域
膠原蛋白增加†
面部張力向量顯著增加
達2.2mm提升效果‡
上市後臨床跟進研究
安全性及效果獲得確認**
安全性與風險管理
良好的風險效益比
HArmonyCa™美神針™的長期安全跟進研究顯示,沒有發生與療程相關的持續不良事件。
常見的副作用包括紅腫、疼痛、觸痛和痕癢,普遍會在24至48小時內得到改善,而腫脹則會在大約一周內完全消退。
療程前
- • 醫生全面評估療程記錄
- • 定制專屬療程方案
- • 評估疼痛管理需求
療程後
- • 可能出現輕微紅腫
- • 即時顯著提升效果
- • 第一週開始膠原新生
常見問題 Q&A
References:
* Results from a prospective and non-randomised interventional study where HArmonyCa™ was injected in the preauricular region of 15 women. The primary endpoint were the volumetric changes at Day 180. Secondary endpoints included changes in facial tension vectors, time to filler tissue integration and safety profile.5
† Ultrasound and elastography examinations were performed using the Samsung HT 30 ultrasound machine and the ElastoScan HS30/XH30 respectively (Samsung Healthcare Global). When compared to baseline, an increase in density, viscoelasticity, and firmness of the tissue was observed at 60 days and then confirmed at 90 days after treatment. At 180 days after treatment an increase in collagen in the treated areas was confirmed.5
‡ Vectra® H2 (Canfield Scientific, Inc.) was used to assess skin lift and tightening by measuring the changes in FTV at 60, 90, and 180 days after treatment. As compared to pre-treatment values, FTV significantly increased at all timepoints with a median (interquartile-range) increased by 2.2 (1.6– 2.2) mm and 2.0 (1.7–2.2) mm in the right and left side, respectively p < 0.0001 each, 180 days after treatment.5
§ HArmonyCa™ can be injected in the preauricular area and in the ascending and descending branches of the jaw.1,5
** A post marketing clinical follow up of safety and performance of HArmonyCa™ (N=62).11
†† Performance evaluation using the 5-point Likert Scale User Satisfaction Questionnaire. Data presented is from the early follow up satisfaction cohort (N=62).11
‡‡ Assessment of general appearance of subjects compared to baseline using the GAIS scale resulted in 91.6 % of subjects graded as showing any improvement. The mean GAIS score for treatment with HArmonyCa™ Lidocaine fell between “improved” and “much improved”, demonstrating clinical effectiveness of the dermal filler (N=48).11
請向醫生或醫學美容中心諮詢切合您獨特需要的專屬療程方案。療程效果因人而異。療程後如有任何不良反應,請即向醫生查詢。
The content on this page is sourced from official clinical studies and manufacturer information for educational and informational purposes only, and should not be used as guidance or reference. Materials are obtained from official sources and online references. If there is any infringement, please contact us for removal.
© 2025 HArmonyCa™. All rights reserved. HK-HAR-250018 | 18/DEC/25



