3大輪廓老化成因 造成疲倦老態


科研實証,97%用家滿意
並樂意推薦6
國際專利技術VYCROSS™
製造,與肌膚組織融合度佳,
效果更自然22
透明質酸
自然存在於人體23
全球110個市場上市24,
安全性備受充分肯定

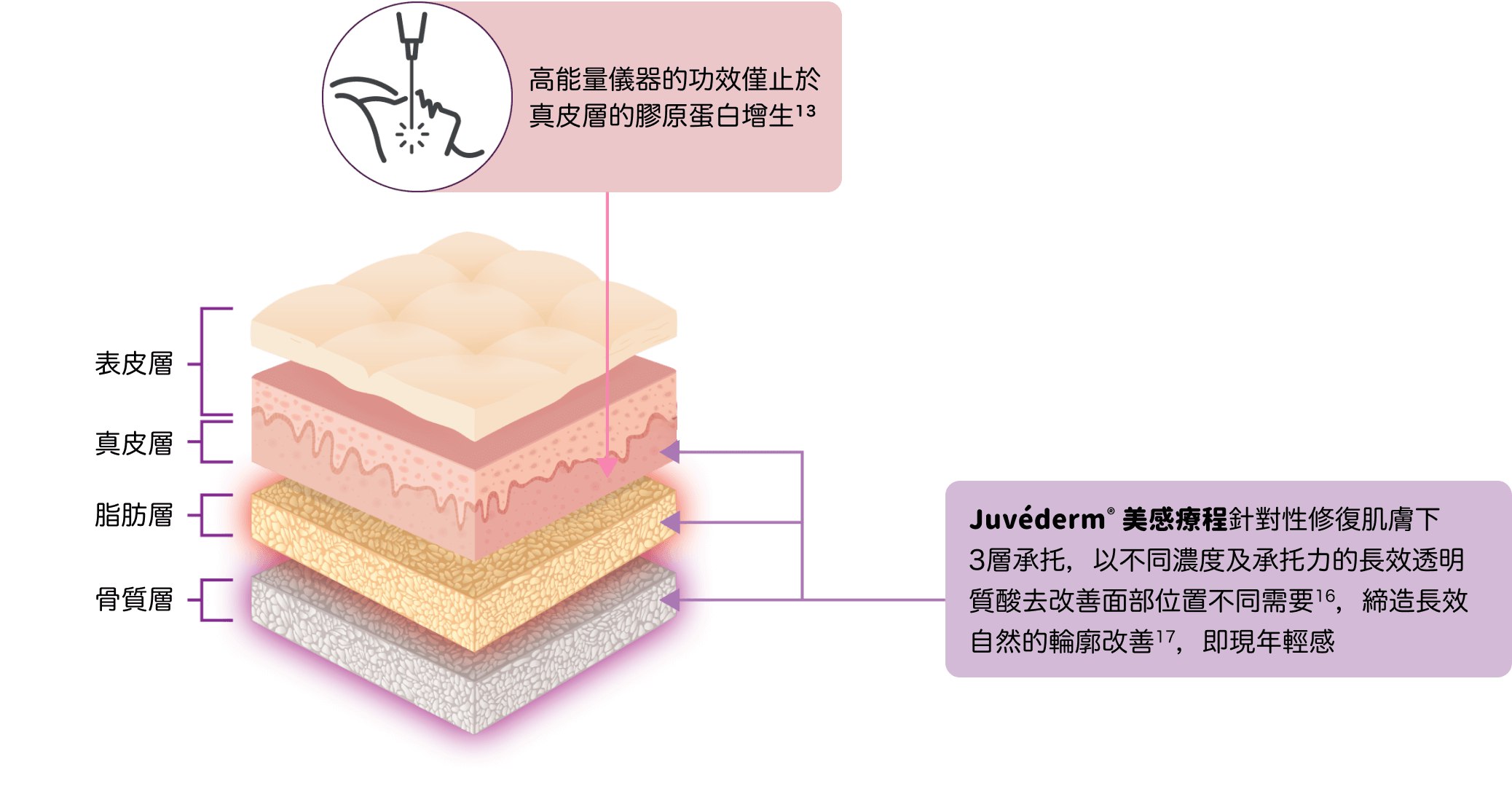
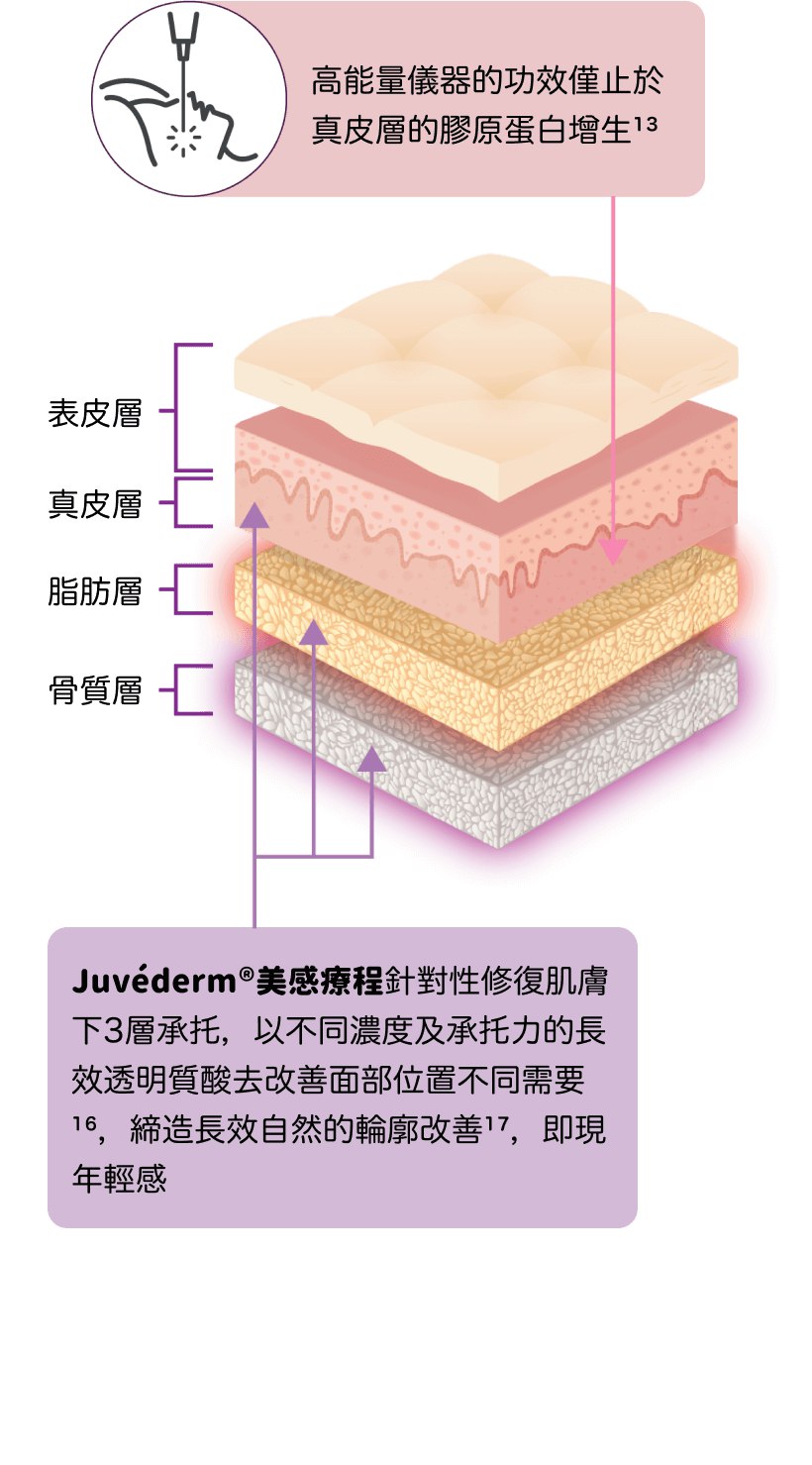
美感療程
提昇輪廓 緊緻豐盈
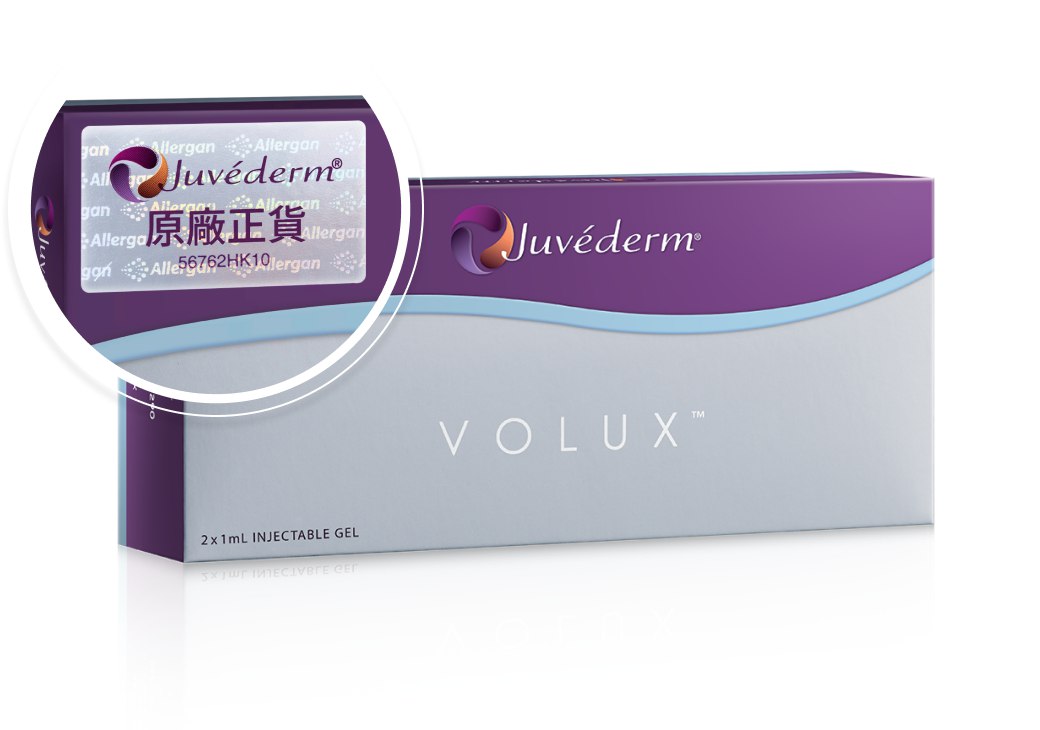
2步認清Juvéderm® 原廠正貨 美麗由你自主

常見問題 Q&A
References:
†Allergan. Unpublished Data. REF-74203. Juvéderm®, the world's leading brand of hyaluronic acid facial fillers. Feb 2019. References: 1. De Maio, M. Aesthetic Plast Surg. 2021 Apr;45(2):690-709; 2. De Maio M, et al. Plast Reconstr Surg. 2017 Aug;140(2):265e-276e; 3. De Maio M, et al. Plast Reconstr Surg. 2017 Oct;140(4):540e-550e; 4. De Maio M. et. al. Plast Reconstr Surg. 2017 Sep;140(3):393e-404e; 6. Callan P et al. Clin Cosmet Investig Dermatol. 2013;6:81-9; 7. Allergan. Data on File. REF-96563. FILLERS GLOBAL NUMBER OF SYRINGES. May 2022. 100 million Juvederm® syringes manufactured worldwide from 2004 to April 2022; 8. VOLUX™, VOLUMA®, VOLIFT®, VOLBELLA® 均獲得歐盟 CE 認證. European Commission, Juvéderm hyaluronic acid based medical devices. EC certificate number: 3824296CE02; 3824296CE06; 3824296CE08; 3824296CE09, Aug 2019; 9. VOLUX™, VOLUMA®, VOLIFT® 及 VOLBELLA® (approval as VOLUX™ XC, VOLUMA® XC, VOLLURE® XC and VOLBELLA® XC in US) 均獲得美國 FDA 認證. U.S. Food and Drug Administration, Department of Health & Human Services. Juvéderm VOLUX XC approval letter P110033/S065, July 2022; Juvéderm VOLUMA XC approval letter P110033/S047, Jun 2020; Juvéderm VOLLURE XC approval letter P110033/S020, Mar 2017; Juvéderm VOLBELLA XC approval letter P110033/S053, May 2021; 10. Papakonstantinou E., et al. Dermatoendocrinol. 2012 Jul 1;4(3):253–258; 11. Tan KS, Oh SR, Priel A, Korn BS, Kikkawa DO (2011). Surgical Anatomy of the Forehead, Eyelids, and Midface for the Aesthetic Surgeon. In Master Techniques in Blepharoplasty and Periorbital Rejuvenation. (pp.11-24). Springer; 12. Mariwalla K. (2011). Rejuvenation of the Upper Face. Medscape Education; 13. Shin JW et al. Int. J. Mol. Sci. 2019,20(9),2126; 16. Goodman GJ et al. Plast Reconstr Surg. 2015;136(5 Suppl):139–48; 17. Allergan. Unpublished Data. INT-JUV-1950127. VYCROSS® – longest duration of hyaluronic acid facial filler in clinical trials. Jan 2020; 18. Jones D and Murphy DK. Dermatol Surg. 2013;39:1602–12. 19. Raspaldo H et al. J Cosmet Dermatol. 2010;9:11–15; 20. Dayan S et al. Aesthet Surg J. 2020 Jun 15;40(7):767-777; 21. Ogilvie P et al. Aesthet Surg J. 2020 Aug 14;40(9):NP499-NP510; 22. Lebreton P. 2004. WO2004092222A2. Cross-linking of low and high molecular weight polysaccharides preparation of injectable monophase hydrogels and polysaccharides and hydrogels thus obtained; 23. De Maio M. Aesthetic Plast Surg. 2004;28:295-300; 24. Allergan. Unpublished Data. REF-80819. Juvéderm®is available in over 110 territories worldwide. Feb 2019; 25. De Maio M, et al. Plast Reconstr Surg. 2017 Aug;140(2):265e-276e; 26. Ogilvie P et al. Aesthet Surg J. 2020 Aug 14;40(9):NP499-NP510; 27: Beer K., et al. Dermatol Surg. 2021. 47(1), 80–85; 28. Ogilvie P et al. Dermatol Surg. 2020 Mar;46(3):376-385; 29. Ecclestron D, Murphy DK. Clin Cosmet Investig Dermatol. 2012;5:167-72; 30. Niforos F et al. Dermatol Surg. 2017;43(10):1271–80; 31. De Maio et. al., Plast Reconstr Surg, 2015 & 2017; 32. Signorini M et al. Plast Reconstr Surg. 2016 Jun;137(6):961e-71e; 33. Urdiales-Galvez F et al. Aesthetic Plast Surg. 2018 Apr;42(2):498-510; 34. Urdiales-Galvez F et al. Aesthetic Plast Surg. 2017 Jun;41(3):667-677; 35. de Almeida AT et al. Surg Cosmet Dermatol 2017;9(3):204-13. 36. Philipp-Dormston WG et al. J Eur Acad Dermatol Venereol. 2017 Jul;31(7):1088-1095; 37. Funt D & Pavicic T. Plast Surg Nurs. 2015 Jan-Mar;35(1):13-32; 38. Shah NS et al. J Am Acad Dermatol. 2002 Aug;47(2):241-4; 39. Fitzgerald R et al. Facial Plast Surg. 2016 Oct;32(5):532-55.
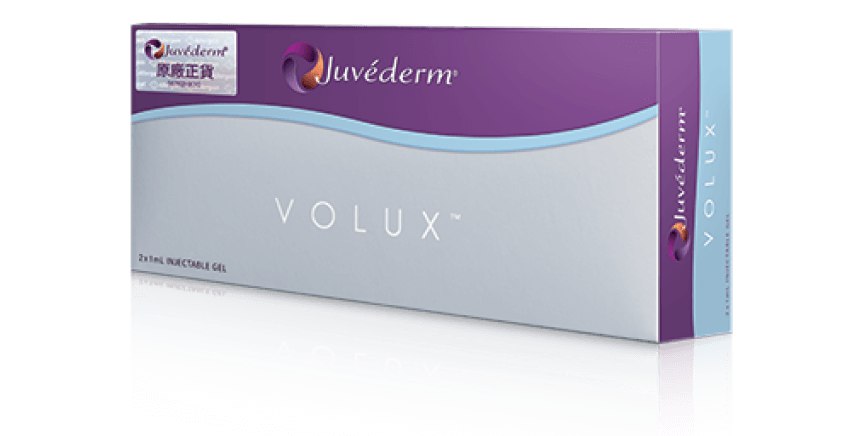
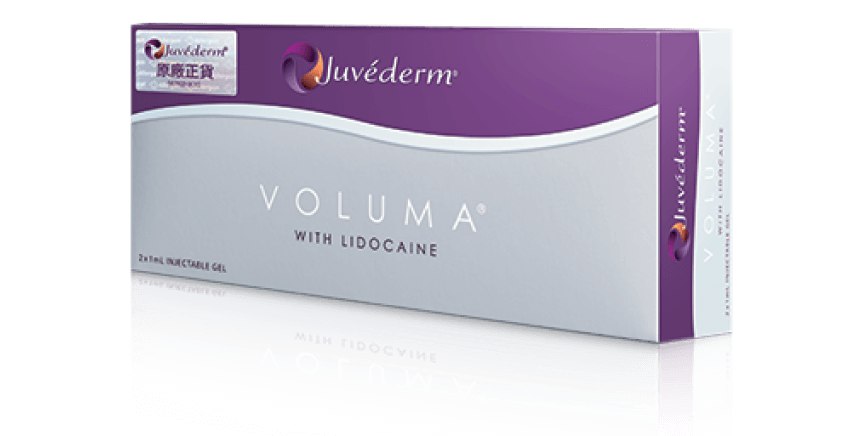
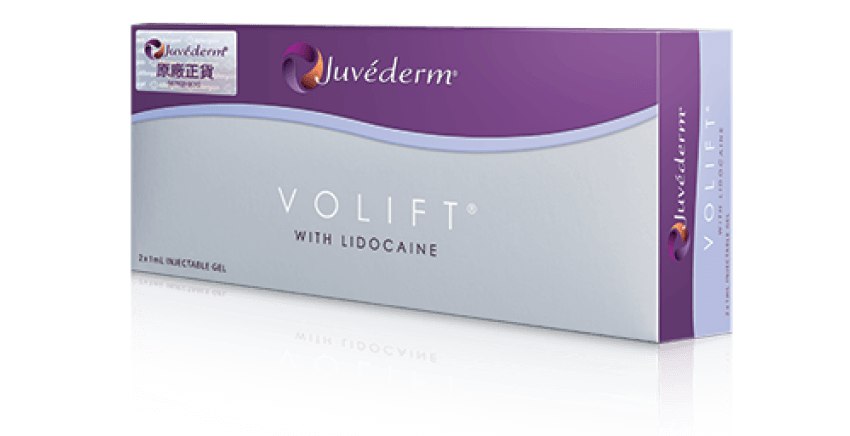
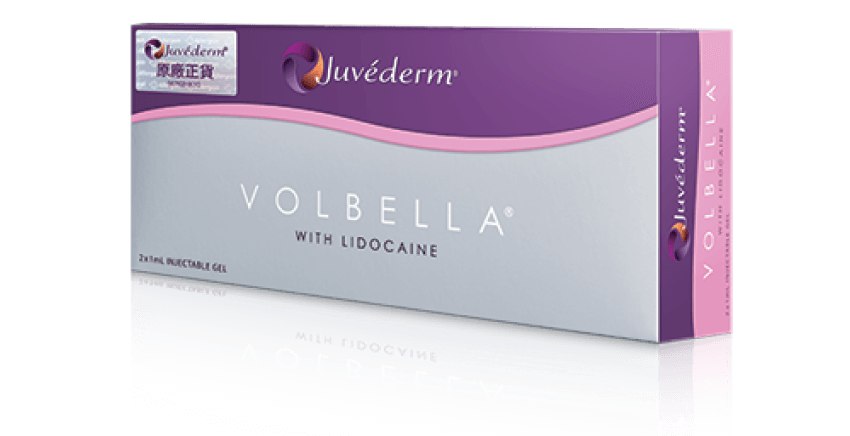
Juvéderm®(Hyaluronic Acid Gel with Lidocaine) 產品系列包括VOLUX™ (HKMD-190341), VOLUMA® (HKMD-100411), VOLIFT® (HKMD-130324), VOLBELLA®(HKMD-210451)。
請向醫生或醫學美容中心諮詢切合您獨特需要的專屬療程方案。療程效果因人而異。療程後如有任何不良反應,請即向醫生查詢。
The content on this page is sourced from Allergan for educational and informational purposes only, and should not be used as guidance or reference. Materials are obtained from Allergan and online sources. If there is any infringement, please contact us for removal.
© 2025 Allergan Aesthetics. All right reserved. HK-JUV-250013 | 25/MAR/25