Restylane® 雙重技術 呈現自然美感





臨床實驗證明,療程效果可長達24個月。Restylane®於瑞典製造,提供多元化的透明質酸療程選擇,配合獨有 NASHA®及 OBT™技術,讓您可以自由搭配,迎合不同美學需要。
Restylane® 產品系列 針對不同需求
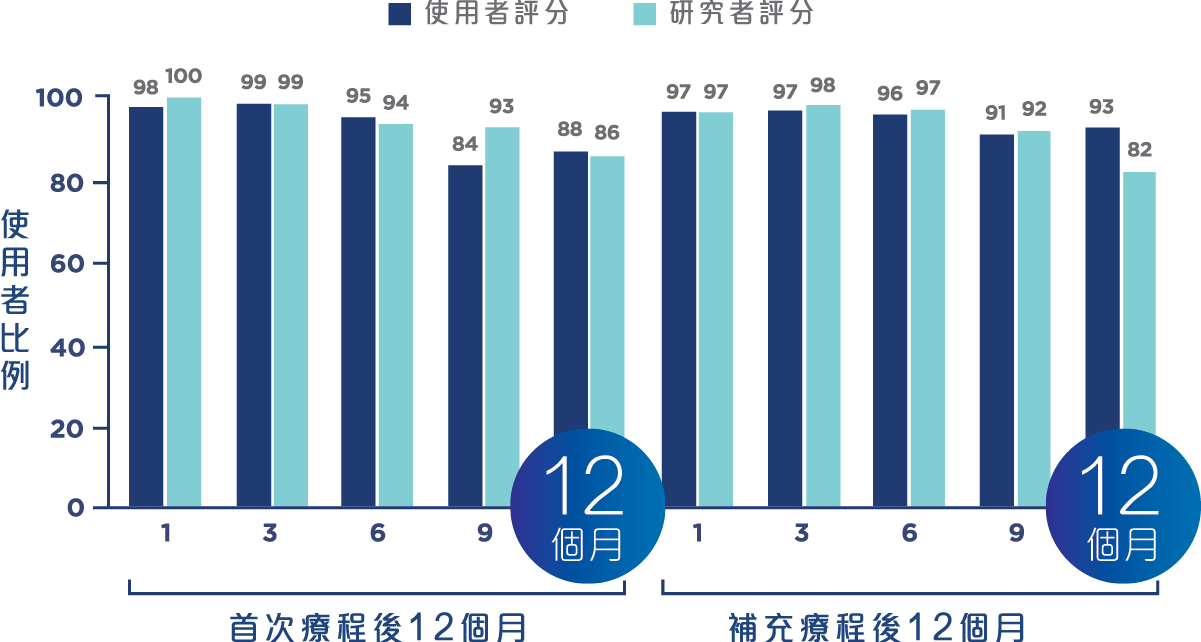
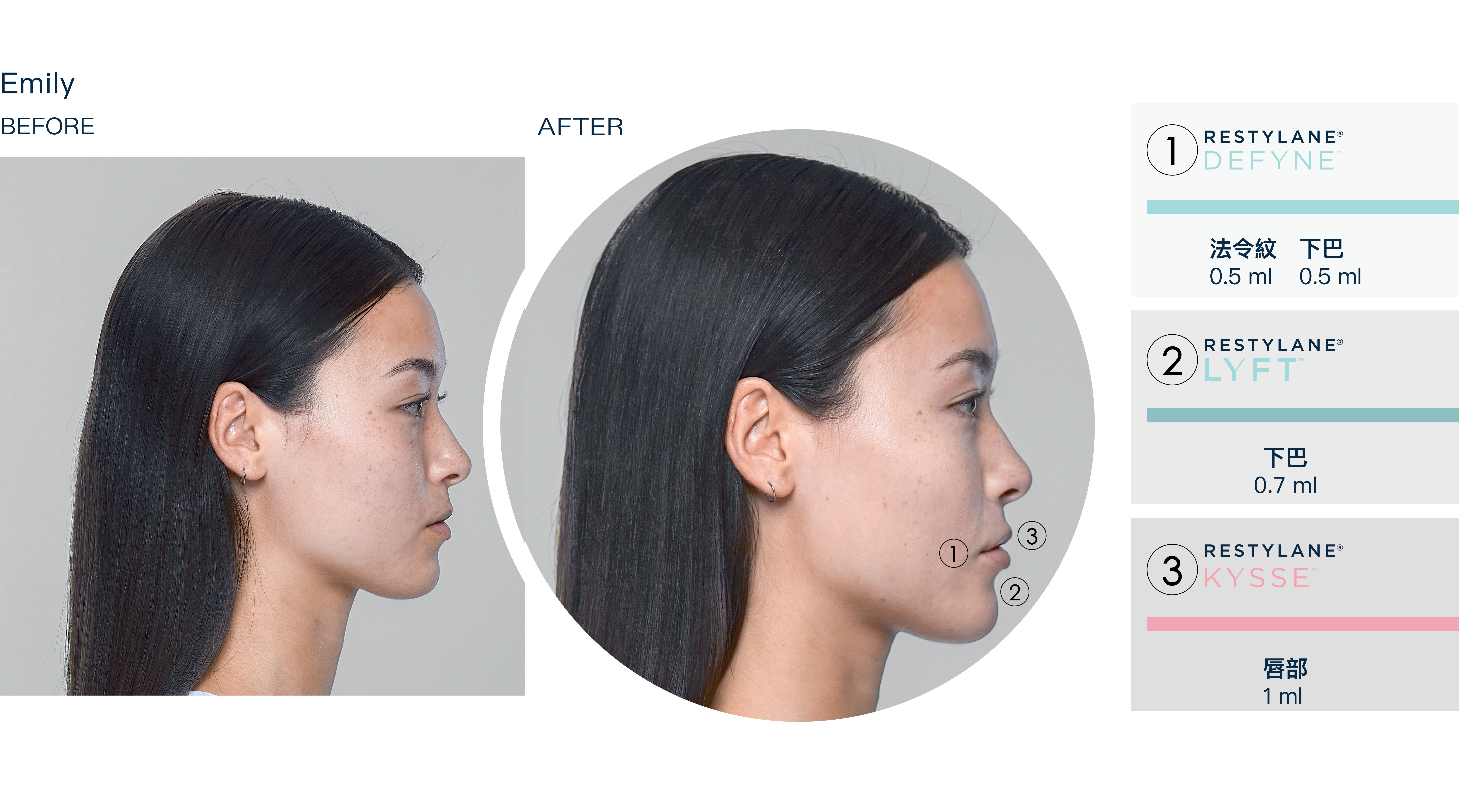
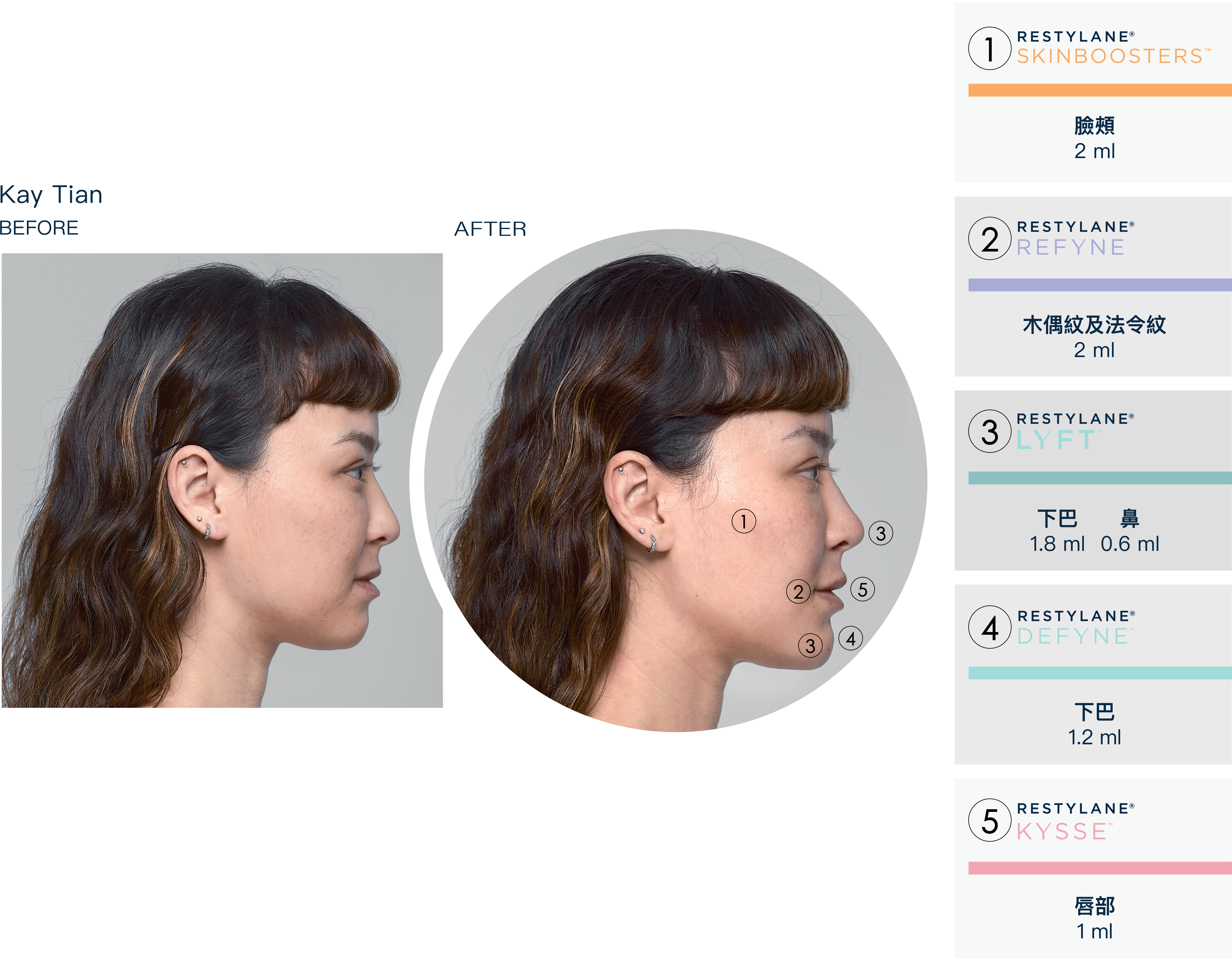
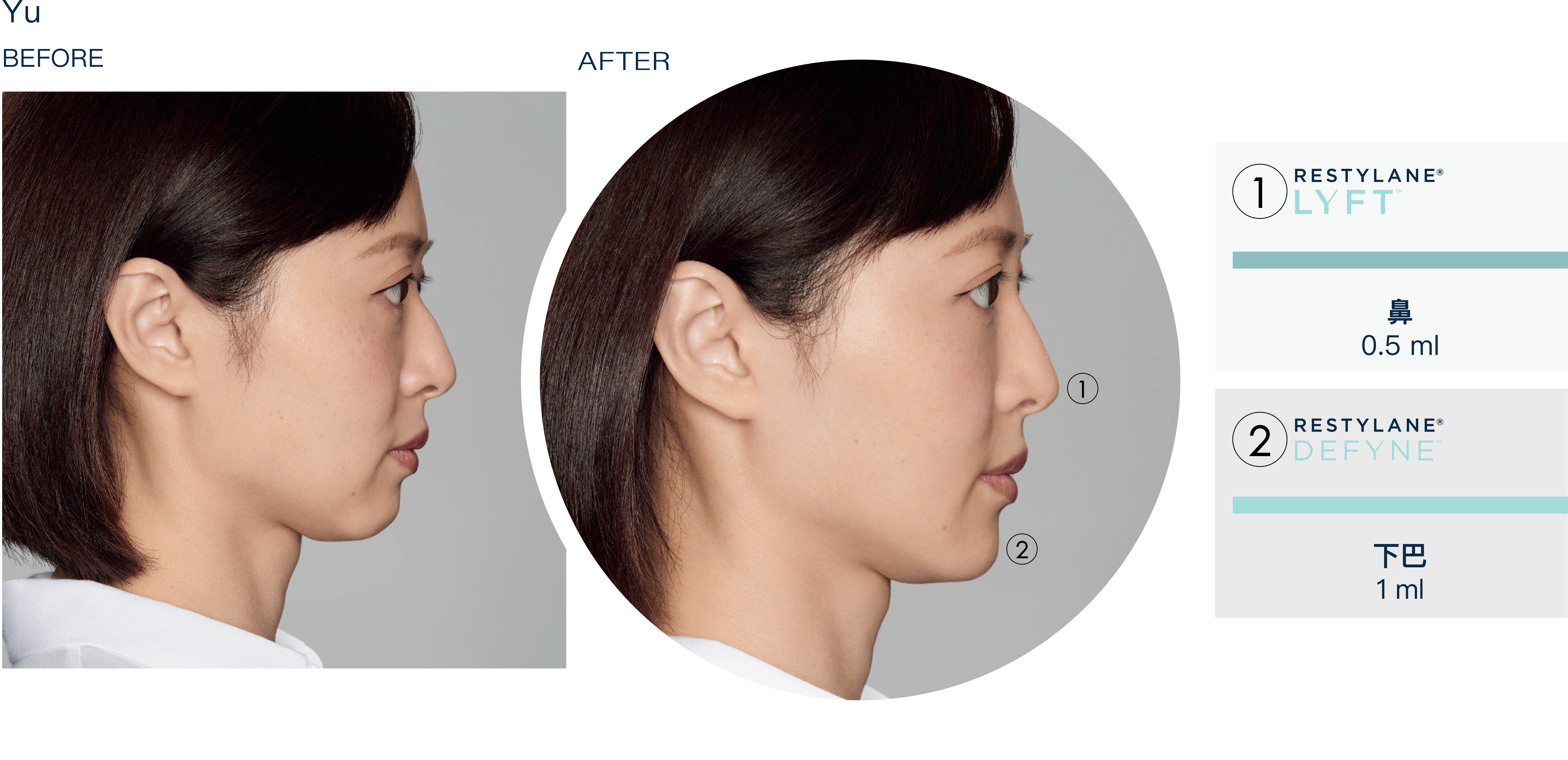
療程效果展示
常見問題 Q&A
References:
1. FDA Summary of safety and effectiveness data. https://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfpma/pma.cfm?id=P040024
2. Restylane® AP37 199679A VEHER*.
3. Ohrlund A. J Cosmet Dermatol. 2018;8:47-54.
4. Kablik J, et al. Dermatol Surg. 2009;35 Suppl 1:302-12.
5. Data on File MA-55607.
6. Data on file MA-57232.
7. Data on file. MA-34483.
8. Data on file. QMS-4933. (Lyft)
9. Data on file. QMS-4706. (Restylane)
10. Data on file. QMS-8662. (Defyne)
11. Data on file. QMS-8665. (Volyme)
12. Data on file. QMS-8668. (Kysse)
13. Data on file. QMS-8633. (Refyne)
14. Andriopoulos B, et al. Presented at: AMWC Monte Carlo, Monaco. 2019.
15. Huang SH, et al. J Drugs Dermatol. 2020;19:836-42.
16. Gregario G et al. Clin Cosmet Investig Dermatol 2022;15: 681-690.
17. Huang S and Tsai T. J Drugs Dermatol 2020;19(9):836-842.
18. Lundgren B et al. J Drugs Dermatol 2018; 17(9): 982-986.
19. Talarico S, et al. Dermatol Surg. 2015;41:1361-9.
20. RESTYLANE® SKINBOOSTERS™ Vital EU IFU.
21. RESTYLANE® SKINBOOSTERS™ Vital Light EU IFU.
22. RESTYLANE® VOLYME™ EU IFU.
23. RESTYLANE® LYFT™ EU IFU.
24. RESTYLANE® EU IFU.
25. RESTYLANE® DEFYNE™ EU IFU.
26. RESTYLANE® REFYNE™ EU IFU.
27. RESTYLANE® KYSSE™ EU IFU.
28. Swift A et al. Clin Cosmet Investig Dermatol 2017;10:229-238.
29. Data on file. MA-48544.
30. Rzany B, Cartier H, Kestemont P, et al. Dermatol Surg. 2012;38:1153-1161.
31. Data on file. MA-39680; Data on file. MA-40221; Data on file. MA-39613.
Restylane® (透明質酸凝膠注射劑) 已獲得歐盟CE及美國FDA認證,全球超過110個市場認可的安全有效美容療程。
請向醫生或醫學美容中心諮詢切合您獨特需要的專屬療程方案。療程效果因人而異。療程後如有任何不良反應,請即向醫生查詢。
The content on this page is sourced from Allergan for educational and informational purposes only, and should not be used as guidance or reference. Materials are obtained from Allergan and online sources. If there is any infringement, please contact us for removal.
© 2025 Galderma HK Limited. All rights reserved. HK-RES-2400032 / July-2024